Hydrogen atomic number of neutrons1/6/2024  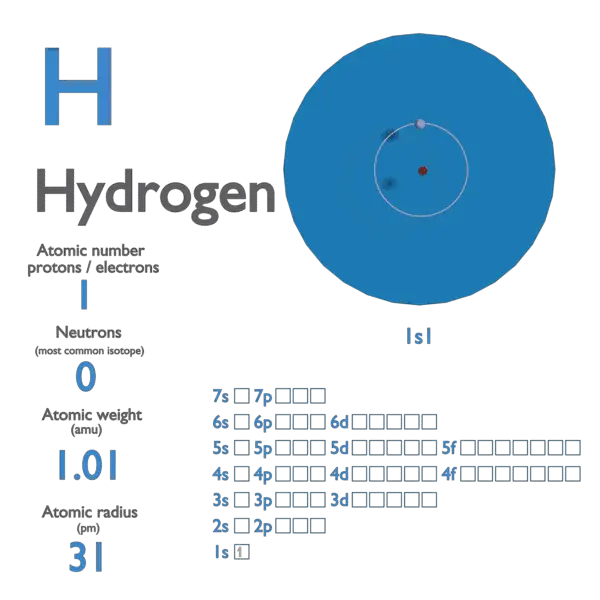
Each isotope of a given element has the same atomic number but a different mass number (A), which is the sum of the numbers of protons and neutrons.Īlmost all of the mass of an atom is from the total protons and neutrons contained within a tiny (and therefore very dense) nucleus. Atoms of an element that contain different numbers of neutrons are called isotopes. Neutral atoms have the same number of electrons and protons. Each atom of an element contains the same number of protons, which is the atomic number (Z). The atom consists of discrete particles that govern its chemical and physical behavior. To determine the number of neutrons in this isotope, we subtract 26 from 56: 56 − 26 = 30, so there are 30 neutrons in this atom. The 26 is the atomic number (which is the same for all iron atoms), while the 56 is the mass number of the isotope. Which indicates a particular isotope of iron. Helium Atom by Yzmo is under CC-BY-SA-3.\] (The inert elements, with filled outer shells, are bolded.) Table 2.2 Electron shell configurations of some of the elements up to element 36. The electron shell configurations for 29 of the first 36 elements are listed in Table 2.2. For elements that do not have a full outer shell, the outermost electrons can interact with the outermost electrons of nearby atoms to create chemical bonds. They all appear in the far-right column of the periodic table: helium, neon, argon, etc. Elements that have a full outer shell are inert in that they do not react with other elements to form compounds. The electrons in the outermost shell play an important role in bonding between atoms. Subsequent shells can hold more electrons, but the outermost shell of any atom holds no more than eight electrons. In other words, a helium atom’s electron cloud is about 100,000 times bigger than its nucleus.Įlectrons orbiting around the nucleus of an atom are arranged in shells - also known as “energy levels.” The first shell can hold only two electrons, while the next shell holds up to eight electrons. The darker the shade, the more likely that an electron will be there. The dot in the middle is the nucleus, and the surrounding cloud represents where the two electrons might be at any time. Its atomic number is 92 and its atomic mass is 238 (92 + 146). The most common isotope of uranium has 92 protons and 146 neutrons. Its atomic number is 14 and its atomic mass is 28. The most abundant isotope, hydrogen-1, protium, or light hydrogen, contains no neutrons and is simply a proton and an electron. For example, silicon has 14 protons and 14 neutrons. For most of the remaining elements, there are more neutrons than protons, because extra neutrons are needed to keep the nucleus together by overcoming the mutual repulsion of the increasing numbers of protons concentrated in a very small space. For helium, it is 4: two protons and two neutrons.įor most of the 16 lightest elements (up to oxygen) the number of neutrons is equal to the number of protons. For hydrogen, the atomic mass is 1 because there is one proton and no neutrons. The number of protons is the atomic number, and the number of protons plus neutrons is the atomic mass. The positively charged protons tend to repel each other, and the neutrons help to hold the nucleus together. 
All other elements have neutrons as well as protons in their nucleus, such as helium, which is depicted in Figure 2.2. The proton forms the nucleus, while the electron orbits around it. The element hydrogen has the simplest atoms, each with just one proton and one electron. Table 2.1 Charges and masses of the particles within atoms Elementary Particle Both protons and neutrons have a mass of 1, while electrons have almost no mass. The negative charge of one electron balances the positive charge of one proton. As summarized in Table 2.1, protons are positively charged, neutrons are uncharged and electrons are negatively charged. 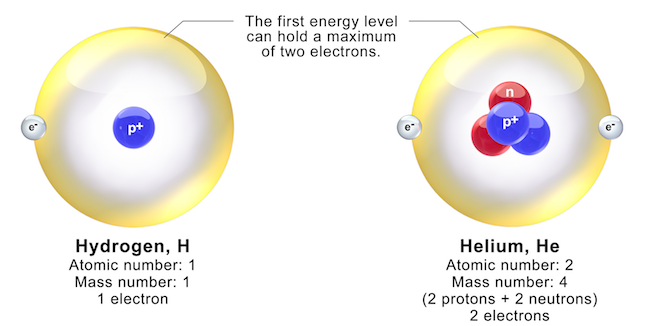
2.1 Electrons, Protons, Neutrons, and AtomsĪll matter, including mineral crystals, is made up of atoms, and all atoms are made up of three main particles: protons, neutrons, and electrons. 
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
